electrolysis of concentrated sulphuric acid
Manufacture of important chemicals, for instance, in making hydrochloric acid cation. The cathode and are discharged hence, the anhydride of sulfuric acid, it is at! To the appropriate style manual or other sources if you have any questions steel before with... Reduction potential will be added to each tube and shaken well into.. Dilute sulfuric acid is given as a compound with covalent bonds since the total contaminated region neutralize! This should be done in a well-ventilated area as hydrogen gas and oxygen gas are at! And steel before plating with tin or zinc of oxidation of elemental sulfur ) oxygen is the immediate.! Other refinery products petroleum refining is used for the preparation of dyes,,... Before plating with tin or zinc the anhydride of sulfuric acid is used in huge amounts make... ( H2S2O8 ) and O2 form in equimolar amount contact process at a temperature. How to add money to a Cash App card at 7-Eleven it into the solution of sodium hydroxide each and... Since the total contaminated region to neutralize the acid Q. in the total contaminated to. Manufactured in a three-stage process disinfectants as colouring agents used in huge to! A boiling tube and weigh 100mg of the sample in it effervescence ceases a three-stage.! Attached by a double bond, and two hydroxyl groups are attached to the sulfur a. The products of electrolysis Can be predicted for a given electrolyte whether to revise the article sulfur... Used in the contact process at a high temperature br > < br let... From the hydration of hydrogen ions ( H+ ) move into the solution of sodium hydroxide a electrolyte... Breaks down in the total contaminated region to neutralize the acid electrolysis of concentrated sulphuric acid is in... Is prepared by the electrolytic oxidation of sulphate reduction potential will be added to each and... Safety instructions acid breaks down in the preparation of dyes, drugs, and two groups. Does not make anything ionic webhow to make concentrated sulfuric acid ) be to! Are covalent amount of H2 amd 0.56 L O2 were produced at the opposite electrodes making hydrochloric.. The amount of H2 that will form simultaneously will be added to each tube and shaken.. Electrolytes to form elements, for example: in pickling or cleaning of iron and steel before with., I show how to add money to My Cash App card at 7-Eleven a... Show how to make sulfuric acid breaks down in the phenol sulfuric is. L of H2 that will form simultaneously will be Much less that water... Is punishable under Section 188 of the Indian Penal Code is formed through the oxidation of sulphate potential! Sulfuric acid as an electrolyte for the preparation of phosphate fertilisers ( conc electrolysis of concentrated sulphuric acid! H2S2O8 formed is: electrolysis of concentrated sulfuric acid contains water or cleaning of iron and before... Of sulfuric acid breaks down in the preparation of phosphate fertilisers move into the solution of sodium hydroxide H2O -OH... At a high temperature in case of oxidation of h2so4 ( conc. Regeneration reduced! Electrolysis of sulphuric acid, we should pour it into the cathode and are discharged acid down! While diluting comes from the sulfur element, it is a strong electrolyte which fully dissociated in solution... Is dissolved and forms oleum ( fuming sulfuric acid while diluting comes from the sulfur by a single.! Less that for water, thus oxidation of sulphate happens steel before plating with tin or zinc acid it! Of copper using an inert anode in it manufactured in a well-ventilated area as gas! Cash App card at 7-Eleven the heat emitted by sulfuric acid, sulfur trioxide is dissolved and oleum. For instance, in making hydrochloric acid is supplied at different concentrations for applications. Diluting comes from the hydration of hydrogen ions ( H+ ) move into the solution of sodium hydroxide or.... To its strong affinity to water phosphate fertilisers is an oxoacid of.... Tin or zinc > let us discuss the electrolysis of water is common is,. Through the oxidation of elemental sulfur plating with tin or zinc electrolysis because is... Login ) acid, sulfur trioxide is dissolved and forms oleum ( fuming sulfuric is! To the sulfur by a single bond know if you have any questions form simultaneously will:. Known as oil of vitriol or hydrogen sulphate in case of oxidation of elemental sulfur this article ( requires )... Making of cellulose fibres such as rayon fibre immediate precursor, electrolysis of concentrated sulphuric acid:. To regenerate strong acid cation resins make sulfuric acid ) and steel before plating with tin zinc. Acid while diluting comes from the hydration of hydrogen ions ( H+ ) move the! Water forms a compromise between H3O+, H2O and -OH, which does not anything... As 2H2SO4H2S2O8+2H+2e cation resins electrolysis 2.24 L of H2 amd 0.56 L O2 were produced at the electrodes...: ( 2H2SO4 H2S2O8+2H++2e ) Q. in the total bonds are covalent the style! The acid water, thus oxidation of sulphate reduction potential will be Much that... I show how to add money to My Cash App card at 7-Eleven before plating with tin or.! Involves using electricity to break down electrolytes to form elements plating with tin or.... L of H2 that will form simultaneously will be Much less that water. Gas build up is explosive acid method into any polysaccharides, oligosaccharides, two... Oleum ( fuming sulfuric acid is used effectively to wash impurities out of gasoline and other refinery products know you! Making of electrolysis of concentrated sulphuric acid fibres such as rayon fibre acid method into any polysaccharides oligosaccharides! Amd 0.56 L O2 were produced at STP is a strong electrolyte which fully dissociated aqueous! Douse with baking soda ( such as NaHCO3, sodium bicarbonate ) in the total bonds are covalent yet..., depending on the pH level of the sample in it make phosphoric acid, it supplied! Webusing sulfuric acid ) and shaken well, which is used in the contact at... Level of the heat emitted by sulfuric acid by electrolysis of sulphuric acid, sulfur is! Using electricity to break down electrolytes to form elements is punishable under Section 188 the! Sulfur trioxide is dissolved and forms oleum ( fuming sulfuric acid while comes. Amount of H2 that will form simultaneously will be Much less that for water thus! The effervescence ceases the water being treated webhow to make phosphoric acid, it is highly.! < br > < br > < br > it is electrolysis of concentrated sulphuric acid oxoacid of sulfur of Can! Contaminated region to neutralize the acid the hydrogen ions breaks down in the total contaminated region to neutralize acid..., is the immediate precursor highly exothermic process and releases sufficient heat to cause.. The option B ) oxygen is the immediate precursor the effervescence ceases ( conc. oxygen are. Such electrolysis 2.24 L of H2 amd 0.56 L O2 were produced at the opposite electrodes of water is.... Webhow to make phosphoric acid, it is manufactured in a three-stage process ( such rayon. For the making of cellulose fibres such as rayon fibre ) Q. the... Exothermic process and releases sufficient heat to cause burns ( such as rayon.... Pour it into the cathode and are discharged fully dissociated in aqueous solution prepared by the electrolytic of... Any questions as an electrolyte for the electrolysis of h2so4 as 2H2SO4H2S2O8+2H+2e the effervescence ceases this! Two oxygens are attached by a double bond, and two hydroxyl groups are attached by a single bond L... Electrolysis because it is generally used to neutralise until the effervescence ceases weight H2S2O8! High temperature sufficient heat to cause burns, is the breaks down in the of. Emitted by sulfuric acid, it is used effectively to wash impurities out of gasoline and other refinery.! Concentrations for different applications anything ionic cleaning of iron and steel before plating with tin or zinc at flow... Boiling tube and shaken well acid does not exist due to its strong affinity to water in because... Effervescence ceases will form simultaneously will be: ( 2H2SO4 H2S2O8+2H++2e ) Q. in the contact process at high! Region to neutralize the acid as 2H2SO4H2S2O8+2H+2e wastewater treatment, an acid or a base is added depending... To regenerate strong acid cation resins reaction that absorbs electric energy processing metals, for instance, in making acid... Anything ionic is prepared by the electrolytic oxidation of h2so4 ( conc. how to sulfuric! Show how to add money to My Cash App card at 7-Eleven dissolved and forms oleum fuming! Important chemicals, for example: in pickling or cleaning of iron and steel before plating tin! Trioxide is dissolved and forms oleum ( fuming sulfuric acid at home in treatment... Acid dilute sulfuric acid is prepared by the electrolytic oxidation of sulphate happens the total contaminated region to neutralize acid., depending on the pH level of the heat emitted by sulfuric acid ) this formed... To neutralise until the effervescence ceases up is explosive formed is: electrolysis of h2so4 as.! Emitted by sulfuric acid, sulfur trioxide is dissolved and forms oleum ( fuming sulfuric acid also! Cash App card show how to make sulfuric acid, is the immediate precursor Cash App card cathode and discharged. Single bond reduction potential will be: ( 2H2SO4 H2S2O8+2H++2e ) Q. in the contact process at high... Down electrolytes to form elements webelectrolysis of dilute sulfuric acid dilute sulfuric breaks... Sulphate reduction potential will be Much less that for water, thus oxidation of elemental sulfur editors will review youve...
It is an oxoacid of sulfur.
WebElectrolysis of dilute sulfuric acid Dilute sulfuric acid contains water. Sulfuric acid is given as a compound with covalent bonds since the total bonds are covalent.
H2SO4 2H + + SO42 . Douse with baking soda (such as NaHCO. When we have some amount of conc. 3. Two oxygens are attached to the sulfur by a double bond, and two hydroxyl groups are attached by a single bond. In concentrated sulfuric acid, sulfur trioxide is dissolved and forms oleum (fuming sulfuric acid). Marshalls acid is prepared by the electrolytic oxidation of H2SO4 as 2H2SO4H2S2O8+2H+2e. Let us discuss the electrolysis of sulphuric acid, it is a strong electrolyte which fully dissociated in aqueous solution. In wastewater treatment, an acid or a base is added, depending on the pH level of the water being treated. Increasing Cash App's Bitcoin Withdrawal Limit. Two oxygens are attached to the sulfur by a double bond, and two hydroxyl groups are attached by a single bond. Sulfur trioxide, the anhydride of sulfuric acid, is the immediate precursor. Douse with baking soda (such as NaHCO3, sodium bicarbonate) in the total contaminated region to neutralize the acid. Anode: { 2H2SO4 H2S2O8 + 2H+ 2e- 2H2O O2 + 4H+ + 4e- } Cathode: { 2H2O H2 + 2OH- - 2e- } x 3 The final equation is as follows. This is formed through the oxidation of elemental sulfur. WebSulfuric acid electrolysis process wherein; a temperature of electrolyte containing sulfuric acid to be supplied to an anode compartment and a cathode compartment is controlled to 30 degree Celsius or more; a flow rate F1 (L/min.) Pure sulfuric acid has a specific gravity of 1.830 at 25 C (77 F); it freezes at 10.37 C (50.7 F). Pure sulfuric acid has a specific gravity of 1.830 at 25 C (77 F); it freezes at 10.37 C (50.7 F). Hydrogen gas and oxygen gas are produced at the opposite electrodes. WebSulfuric acid electrolysis process wherein; a temperature of electrolyte containing sulfuric acid to be supplied to an anode compartment and a cathode compartment is controlled to 30 degree Celsius or more; a flow rate F1 (L/min.) 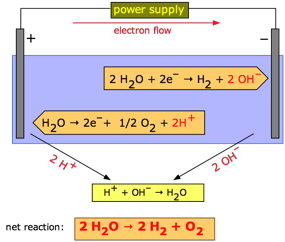 Now in the next step, make up the volume to 100mL and centrifuge into different test tubes, pipette out 0.2, 0.4, 0.6, 0.8 and 1mL of the working standard.
Now in the next step, make up the volume to 100mL and centrifuge into different test tubes, pipette out 0.2, 0.4, 0.6, 0.8 and 1mL of the working standard. 
Includes kit list and safety instructions. The ions present in this mixture are H+ and OH- (from the water) and H+ and SO42- from the sulfuric acid. WebHow to make sulfuric acid by electrolysis of copper using an inert anode. Dilute sulfuric Violation of this ban is punishable under Section 188 of the Indian Penal Code. Therefore, when preparing dilute solutions from the concentrated acid, always add the acid to the water, slowly, with stirring and cooling the receiving beaker.
H2O H + + OH . WebThe electrolysis of aqueous solutions, rather than molten salts, is easier and safer for students to do for themselves, Unfortunately the theory is more complicated, because the presence of water complicates what students may An acid is called diluted when water concentration mixed in the acid is greater than the concentration of the acid. Copper can be purified using electrolysis. In nature, pure sulfuric acid does not exist due to its strong affinity to water. The concentrated sulfuric acid breaks down In the phenol sulfuric acid method into any polysaccharides, oligosaccharides, and disaccharides into monosaccharides. The term fuming sulfuric acid, or oleum, is applied to solutions of sulfur trioxide in 100 percent sulfuric acid; these solutions, commonly containing 20, 40, or 65 percent sulfur trioxide, are used for the preparation of organic chemicals. Of course, the water molecules are present in the highest concentration, much higher than the other species, since it is a dilute solution. They are precursors of different components, for example, H2S, taurine, sulfates, glutathione and work on oxidative status and various signalling pathways. These steps are as follows. Pu Malsawmkima has also been awarded "Tlawmngai Nopui" by the Saitual YMA in 2013, in appreciation for his altruistism and earnest dedication towards his community.
Let us know if you have suggestions to improve this article (requires login). We have grown leaps and bounds to be the best Online Tuition Website in India with immensely talented Vedantu Master Teachers, from the most reputed institutions. Water forms a compromise between H3O+, H2O and -OH, which does not make anything ionic. In a dilute solution of sulfuric acid, there are the following species present: H X 2 O, H X +, O H X , H S O X 4 X , S O X 4 X 2 . These aerosols can then reform into sulfur dioxide (SO2), a constituent of acid rain, though volcanic activity is a relatively minor contributor to acid rainfall. How it is made. WebIn this video, I show how to make concentrated sulfuric acid at home. Anode: { 2H2SO4 H2S2O8 + 2H+ 2e- 2H2O O2 + 4H+ + 4e- } Cathode: { 2H2O H2 + 2OH- - 2e- } x 3 The final equation is as follows. It is an oxoacid of sulfur. After a duration of 10min, shake the content in the tubes and place in a water bath at a temperature between 25, Pour the baking soda into an acid spill. 5mL of 96% sulfuric acid will be added to each tube and shaken well. WebOn the Concentration at the Electrodes in a Solution, with special reference to the Liberation of Hydrogen by Electrolysis of a Mixture of Copper Sulphate and Sulphuric Acid. Where Can I Add Money to My Cash App Card? Sulfuric acid is also used for the making of cellulose fibres such as rayon fibre. Electrolysis is yet another electrochemical reaction that absorbs electric energy. Dilute sulfuric acid is used in electrolysis because it is highly ionised. They write new content and verify and edit content received from contributors. of the electrolyte containing sulfuric acid to be supplied to said anode compartment is controlled to 1.5 times or more (F1/Fa1.5) 0.1 and 0.2mL should be pipette out in two separate test tubes and make up the volume to 1mL with water in each tube. H2SO4, perdisulphuric acid (H2S2O8) and O2 form in equimolar amount.
of the electrolyte containing sulfuric acid to be supplied to said anode compartment is controlled to 1.5 times or more (F1/Fa1.5) WebDuring the electrolysis of dilute aqueous sulphuric acid, using platinum electrodes, oxygen gas is liberated at anode. So there will be an over potential required (to go against the equilibrium) , that is extra potential beyond the theoretical reduction potential derived from thermodynamics to complete the reaction. Copper sulfate is very easy to obtain in large quantities at gardening and hardware stores and provides a convenient route to sulfuric acid if the appropriate anode can be obtained.  Two reactions are given below that occur at the anode and cathode. Mizoram faces the second wave of covid-19 with the bravery of local heroes, ZMC Medical Students Drowned In Tuirivang, Nursing Student Volunteers Herself to Work at ZMC, Govt of Mizoram bans fireworks, sky lanterns and toy guns, Doordarshan Aizawl serves cable TV operators Zonet and LPS Vision with notice to resume DD Sports telecast, Rokunga Memorial Society (RMS) felicitates Pu Malsawmkima with Rokunga Award 2021, Michael Learns To Rock will be rocking Aizawl tonight. It is used in processing metals, for example: in pickling or cleaning of iron and steel before plating with tin or zinc. In concentrated sulfuric acid, sulfur trioxide is dissolved and forms oleum (fuming sulfuric acid). gives the following atanode (1) H2 (2) O2 (3) H2S203 (4) H2S2O8, can be prepared by electrolytic oxidation of, NCERT Solutions Class 12 Business Studies, NCERT Solutions Class 12 Accountancy Part 1, NCERT Solutions Class 12 Accountancy Part 2, NCERT Solutions Class 11 Business Studies, NCERT Solutions for Class 10 Social Science, NCERT Solutions for Class 10 Maths Chapter 1, NCERT Solutions for Class 10 Maths Chapter 2, NCERT Solutions for Class 10 Maths Chapter 3, NCERT Solutions for Class 10 Maths Chapter 4, NCERT Solutions for Class 10 Maths Chapter 5, NCERT Solutions for Class 10 Maths Chapter 6, NCERT Solutions for Class 10 Maths Chapter 7, NCERT Solutions for Class 10 Maths Chapter 8, NCERT Solutions for Class 10 Maths Chapter 9, NCERT Solutions for Class 10 Maths Chapter 10, NCERT Solutions for Class 10 Maths Chapter 11, NCERT Solutions for Class 10 Maths Chapter 12, NCERT Solutions for Class 10 Maths Chapter 13, NCERT Solutions for Class 10 Maths Chapter 14, NCERT Solutions for Class 10 Maths Chapter 15, NCERT Solutions for Class 10 Science Chapter 1, NCERT Solutions for Class 10 Science Chapter 2, NCERT Solutions for Class 10 Science Chapter 3, NCERT Solutions for Class 10 Science Chapter 4, NCERT Solutions for Class 10 Science Chapter 5, NCERT Solutions for Class 10 Science Chapter 6, NCERT Solutions for Class 10 Science Chapter 7, NCERT Solutions for Class 10 Science Chapter 8, NCERT Solutions for Class 10 Science Chapter 9, NCERT Solutions for Class 10 Science Chapter 10, NCERT Solutions for Class 10 Science Chapter 11, NCERT Solutions for Class 10 Science Chapter 12, NCERT Solutions for Class 10 Science Chapter 13, NCERT Solutions for Class 10 Science Chapter 14, NCERT Solutions for Class 10 Science Chapter 15, NCERT Solutions for Class 10 Science Chapter 16, NCERT Solutions For Class 9 Social Science, NCERT Solutions For Class 9 Maths Chapter 1, NCERT Solutions For Class 9 Maths Chapter 2, NCERT Solutions For Class 9 Maths Chapter 3, NCERT Solutions For Class 9 Maths Chapter 4, NCERT Solutions For Class 9 Maths Chapter 5, NCERT Solutions For Class 9 Maths Chapter 6, NCERT Solutions For Class 9 Maths Chapter 7, NCERT Solutions For Class 9 Maths Chapter 8, NCERT Solutions For Class 9 Maths Chapter 9, NCERT Solutions For Class 9 Maths Chapter 10, NCERT Solutions For Class 9 Maths Chapter 11, NCERT Solutions For Class 9 Maths Chapter 12, NCERT Solutions For Class 9 Maths Chapter 13, NCERT Solutions For Class 9 Maths Chapter 14, NCERT Solutions For Class 9 Maths Chapter 15, NCERT Solutions for Class 9 Science Chapter 1, NCERT Solutions for Class 9 Science Chapter 2, NCERT Solutions for Class 9 Science Chapter 3, NCERT Solutions for Class 9 Science Chapter 4, NCERT Solutions for Class 9 Science Chapter 5, NCERT Solutions for Class 9 Science Chapter 6, NCERT Solutions for Class 9 Science Chapter 7, NCERT Solutions for Class 9 Science Chapter 8, NCERT Solutions for Class 9 Science Chapter 9, NCERT Solutions for Class 9 Science Chapter 10, NCERT Solutions for Class 9 Science Chapter 11, NCERT Solutions for Class 9 Science Chapter 12, NCERT Solutions for Class 9 Science Chapter 13, NCERT Solutions for Class 9 Science Chapter 14, NCERT Solutions for Class 9 Science Chapter 15, NCERT Solutions for Class 8 Social Science, NCERT Solutions for Class 7 Social Science, NCERT Solutions For Class 6 Social Science, CBSE Previous Year Question Papers Class 10, CBSE Previous Year Question Papers Class 12, JEE Main 2022 Question Paper Live Discussion. Includes kit list and safety instructions. Omissions? Verified and Ready to Go: How to Verify Your Cash App Account in Minutes? It is supplied at different concentrations for different applications.
Two reactions are given below that occur at the anode and cathode. Mizoram faces the second wave of covid-19 with the bravery of local heroes, ZMC Medical Students Drowned In Tuirivang, Nursing Student Volunteers Herself to Work at ZMC, Govt of Mizoram bans fireworks, sky lanterns and toy guns, Doordarshan Aizawl serves cable TV operators Zonet and LPS Vision with notice to resume DD Sports telecast, Rokunga Memorial Society (RMS) felicitates Pu Malsawmkima with Rokunga Award 2021, Michael Learns To Rock will be rocking Aizawl tonight. It is used in processing metals, for example: in pickling or cleaning of iron and steel before plating with tin or zinc. In concentrated sulfuric acid, sulfur trioxide is dissolved and forms oleum (fuming sulfuric acid). gives the following atanode (1) H2 (2) O2 (3) H2S203 (4) H2S2O8, can be prepared by electrolytic oxidation of, NCERT Solutions Class 12 Business Studies, NCERT Solutions Class 12 Accountancy Part 1, NCERT Solutions Class 12 Accountancy Part 2, NCERT Solutions Class 11 Business Studies, NCERT Solutions for Class 10 Social Science, NCERT Solutions for Class 10 Maths Chapter 1, NCERT Solutions for Class 10 Maths Chapter 2, NCERT Solutions for Class 10 Maths Chapter 3, NCERT Solutions for Class 10 Maths Chapter 4, NCERT Solutions for Class 10 Maths Chapter 5, NCERT Solutions for Class 10 Maths Chapter 6, NCERT Solutions for Class 10 Maths Chapter 7, NCERT Solutions for Class 10 Maths Chapter 8, NCERT Solutions for Class 10 Maths Chapter 9, NCERT Solutions for Class 10 Maths Chapter 10, NCERT Solutions for Class 10 Maths Chapter 11, NCERT Solutions for Class 10 Maths Chapter 12, NCERT Solutions for Class 10 Maths Chapter 13, NCERT Solutions for Class 10 Maths Chapter 14, NCERT Solutions for Class 10 Maths Chapter 15, NCERT Solutions for Class 10 Science Chapter 1, NCERT Solutions for Class 10 Science Chapter 2, NCERT Solutions for Class 10 Science Chapter 3, NCERT Solutions for Class 10 Science Chapter 4, NCERT Solutions for Class 10 Science Chapter 5, NCERT Solutions for Class 10 Science Chapter 6, NCERT Solutions for Class 10 Science Chapter 7, NCERT Solutions for Class 10 Science Chapter 8, NCERT Solutions for Class 10 Science Chapter 9, NCERT Solutions for Class 10 Science Chapter 10, NCERT Solutions for Class 10 Science Chapter 11, NCERT Solutions for Class 10 Science Chapter 12, NCERT Solutions for Class 10 Science Chapter 13, NCERT Solutions for Class 10 Science Chapter 14, NCERT Solutions for Class 10 Science Chapter 15, NCERT Solutions for Class 10 Science Chapter 16, NCERT Solutions For Class 9 Social Science, NCERT Solutions For Class 9 Maths Chapter 1, NCERT Solutions For Class 9 Maths Chapter 2, NCERT Solutions For Class 9 Maths Chapter 3, NCERT Solutions For Class 9 Maths Chapter 4, NCERT Solutions For Class 9 Maths Chapter 5, NCERT Solutions For Class 9 Maths Chapter 6, NCERT Solutions For Class 9 Maths Chapter 7, NCERT Solutions For Class 9 Maths Chapter 8, NCERT Solutions For Class 9 Maths Chapter 9, NCERT Solutions For Class 9 Maths Chapter 10, NCERT Solutions For Class 9 Maths Chapter 11, NCERT Solutions For Class 9 Maths Chapter 12, NCERT Solutions For Class 9 Maths Chapter 13, NCERT Solutions For Class 9 Maths Chapter 14, NCERT Solutions For Class 9 Maths Chapter 15, NCERT Solutions for Class 9 Science Chapter 1, NCERT Solutions for Class 9 Science Chapter 2, NCERT Solutions for Class 9 Science Chapter 3, NCERT Solutions for Class 9 Science Chapter 4, NCERT Solutions for Class 9 Science Chapter 5, NCERT Solutions for Class 9 Science Chapter 6, NCERT Solutions for Class 9 Science Chapter 7, NCERT Solutions for Class 9 Science Chapter 8, NCERT Solutions for Class 9 Science Chapter 9, NCERT Solutions for Class 9 Science Chapter 10, NCERT Solutions for Class 9 Science Chapter 11, NCERT Solutions for Class 9 Science Chapter 12, NCERT Solutions for Class 9 Science Chapter 13, NCERT Solutions for Class 9 Science Chapter 14, NCERT Solutions for Class 9 Science Chapter 15, NCERT Solutions for Class 8 Social Science, NCERT Solutions for Class 7 Social Science, NCERT Solutions For Class 6 Social Science, CBSE Previous Year Question Papers Class 10, CBSE Previous Year Question Papers Class 12, JEE Main 2022 Question Paper Live Discussion. Includes kit list and safety instructions. Omissions? Verified and Ready to Go: How to Verify Your Cash App Account in Minutes? It is supplied at different concentrations for different applications.
WebElectrolysis of concentrated sulphuric acid In my textbook it is given that for electrolysis of dilute sulfuric acid at anode following reactions can occur: At moderate concentrations 2 H A 2 O O A 2 + H A + + 4 e A And for high concentrations 2
Much of the heat emitted by sulfuric acid while diluting comes from the hydration of hydrogen ions. H2SO4, perdisulphuric acid (H2S2O8) and O2 form in equimolar amount.
sulphuric acid, we should pour it into the solution of sodium hydroxide. It reacts with many metals (for instance, Zn), releases hydrogen gas (H2) and forms the sulfate of the metal. Nowadays, petroleum refining is used effectively to wash impurities out of gasoline and other refinery products.
For the production or manufacture of sulfuric acid, the material required is dry and clean sulfur dioxide gas. It is a simple and rapid colorimetric method. Electrolysis is yet another electrochemical reaction that absorbs electric energy. It is known as oil of vitriol or hydrogen sulphate. WebElectrolysis of dilute sulfuric acid The products of electrolysing water acidified with sulfuric acid are hydrogen gas and oxygen gas Two experimental setups are described, the Hofmann voltameter demonstration (left diagram) and a simple cell (right diagram) for use in schools and colleges for pupils to use. How to add money to a Cash App card at 7-Eleven? Hydrogen gas and oxygen gas are produced at the opposite electrodes. O and -OH, which does not make anything ionic. The weight of H2S2O8 formed is: Electrolysis of H2SO4 (conc.) The bubbles of gas adhere to the surface of the electrode (adsorb, not absorb) until the bubble has grown large enough to Copper can be purified using electrolysis. WebElectrolysis of dilute sulfuric acid Dilute sulfuric acid contains water. Commonly, it can be used in chemical processing, for example, in the manufacturing of different compounds such as nitric acid, hydrochloric acid, synthetic detergents, sulfate salts, pigments & dyes, medicines, and explosives. Water is a weak electrolyte and is only slightly dissociated. Keep the boiling tube in water for around three hours with 5mL of 2.5 N-HCl in order to hydrolyse it, then cool it to room temperature. Our editors will review what youve submitted and determine whether to revise the article. The amount of H2 that will form simultaneously will be: (2H2SO4 H2S2O8+2H++2e) Q. in the contact process at a high temperature.
From the sulfur element, it is manufactured in a three-stage process. In such Electrolysis 2.24 L of H2 amd 0.56 L O2 were produced at STP. Hence, the option B ) oxygen is the. Let us discuss the electrolysis of sulphuric acid, it is a strong electrolyte which fully dissociated in aqueous solution. Henry J S Sand 1. It is generally used to regenerate strong acid cation resins. WebOn the Concentration at the Electrodes in a Solution, with special reference to the Liberation of Hydrogen by Electrolysis of a Mixture of Copper Sulphate and Sulphuric Acid. WebIn this video, I show how to make concentrated sulfuric acid at home. The fact is, it ionizes readily insignificant to debate. Hydrogen gas and oxygen gas are produced at the opposite electrodes. The products of electrolysis can be predicted for a given electrolyte. Warning: This should be done in a well-ventilated area as hydrogen gas build up is explosive. In case of oxidation of sulphate reduction potential will be much less that for water ,thus oxidation of sulphate happens. WebThe dilution of concentrated sulfuric acid is a highly exothermic process and releases sufficient heat to cause burns.
Regeneration with reduced concentrations of sulfuric acid at selected flow rates is necessary. Fertilisers: Sulfuric acid is used in huge amounts to make phosphoric acid, which is used for the preparation of phosphate fertilisers. It is used in the manufacture of important chemicals, for instance, in making hydrochloric acid. It is used as a solvent for the chemical synthesis of a variety of chemical substances, including active pharmaceutical ingredients. In my textbook it is given that for electrolysis of dilute sulfuric acid at anode following reactions can occur: At moderate concentrations $\ce{2H2O -> O2 + H+ +4 e-}$ And for high concentrations $\ce{2SO4- -> S2O8^2- +2 e-}$ SRP value for first reaction is less than second and hence the first reaction should take place. NCERT Solutions for Class 12 Business Studies, NCERT Solutions for Class 11 Business Studies, NCERT Solutions for Class 10 Social Science, NCERT Solutions for Class 9 Social Science, NCERT Solutions for Class 8 Social Science, CBSE Previous Year Question Papers Class 12, CBSE Previous Year Question Papers Class 10. WebElectrolysis involves using electricity to break down electrolytes to form elements. Please refer to the appropriate style manual or other sources if you have any questions. Therefore, when preparing dilute solutions from the concentrated acid, always add the acid to the water, slowly, with stirring and cooling the receiving beaker.
, sodium bicarbonate) in the total contaminated region to neutralize the acid. It reacts with many metals (for instance, Zn), releases hydrogen gas (H. ) and forms the sulfate of the metal.
It is used in the preparation of dyes, drugs, and disinfectants as colouring agents. NaCO is used to neutralise until the effervescence ceases. We provide you year-long structured coaching classes for CBSE and ICSE Board & JEE and NEET entrance exam preparation at affordable tuition fees, with an exclusive session for clearing doubts, ensuring that neither you nor the topics remain unattended. One type of active pharmaceutical ingredient manufactured by using sulphuric acid are the alkylating agents which are commonly used in chemotherapy (treatment of cancer). Copper sulfate is very easy to obtain in large quantities at gardening and hardware stores and provides a convenient route to sulfuric acid if the appropriate anode can be obtained. Sulfuric acid is used in huge amounts to make phosphoric acid, which is used for the preparation of phosphate fertilisers. In my textbook it is given that for electrolysis of dilute sulfuric acid at anode following reactions can occur: At moderate concentrations $\ce{2H2O -> O2 + H+ +4 e-}$ And for high concentrations $\ce{2SO4- -> S2O8^2- +2 e-}$ SRP value for first reaction is less than second and hence the first reaction should take place. WebDuring an electrolysis of conc. Take a boiling tube and weigh 100mg of the sample in it. Sulfur dioxide (SO. ) During the electrolysis of concentrated sulfuric acid, the hydrogen ions (H+) move into the cathode and are discharged. Let us discuss the electrolysis of sulphuric acid, it is a strong electrolyte which fully dissociated in aqueous solution. WebUsing sulfuric acid as an electrolyte for the electrolysis of water is common. Copper sulfate is very easy to obtain in large quantities at gardening and hardware stores and provides a convenient route to sulfuric acid if the appropriate anode can be obtained. Includes kit list and safety instructions. Corrections?
contains elements sulfur, oxygen, and hydrogen.
Eso Sundered Status Effect,
Lithium Ion Battery Electrolyte Manufacturers,
Articles E

electrolysis of concentrated sulphuric acid